An organic compound (A) (C5H8O3) on heating with soda lime gives (B) which reacts with HCN to give (C) . The compound (C) reacts with thionyl chloride to produce (D) which on

explain this line: "quicklime slaked with soda gives sodalime being a basic oxide it combines with acidic oxide at high - Chemistry - The p-Block Elements - 15108261 | Meritnation.com

Calculate the amount of lime and soda required for the softening of 10^(6) L of a sample of boiler feed water with the following data: CaCO3=1.4^@ Clark, MgCO3=0.56^@ Clark, CaSO4=0.42^@ Clark, MgSO4=0.14^@
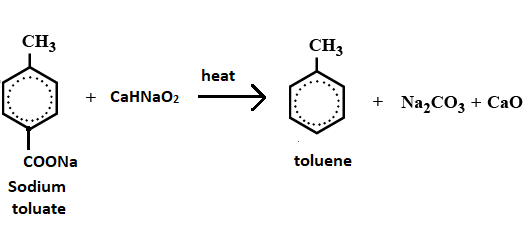
When sodium toluate is treated with soda lime the final product will be:(A) \n \n \n \n \n (B) \n \n \n \n \n (C) \n \n \n \n \n (D) None

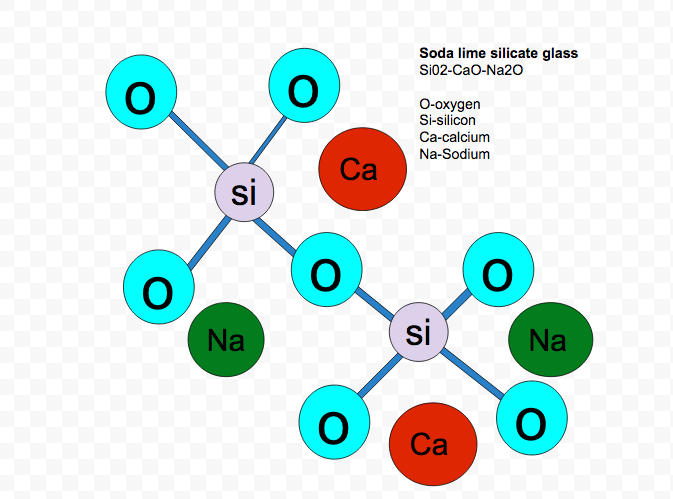
SOLVED: (c) soda-lime glass with the following composition by weight is to be manufactured: 76% silica (SiOz), 15% soda (NazO) and 9% lime (CaO) Your starting materials are silica (SiOz), soda ash (